Gas Solubilities (Physical and Chemical)
Gas solubility data are very important for gas absorption desorption processes like sour gas treatment or carbon capture from flue gas. For gas solubility measurements different methods (synthetic or analytic) from very small partial pressures up to high pressure over a broad range of temperature (- 90 °C up to 400 °C) available. Our laboratory has a broad expertise with gas solubility measurements e.g. in amine solutions where investigations of several dozens of different recipes and compositions where investigated in the past years.
For single gases in liquid solvents the amount of gas in the gas phase is calculated at the given partial (gas) pressure from the known PVT behavior. The partial pressure is obtained during an iterative procedure:
P (gas) = P (system) – x (solvent) × PS (solvent)
For gas mixtures it is usually not possible to calculate the partial pressure for all gases due to the different solubility in the liquids. Thus, a gas phase analysis has to be performed and the partial pressures of all gases are obtained by the following equation,
Pi = yi × P (system)
so that the raw data can then be treated in the usual way to obtain the isothermal P-x data, but no assumptions about the vapor phase are needed.
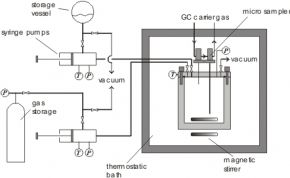
In this case, the experimental procedure is as follows. After filling the cell with a certain amount of liquid and gases, the values for the synthetic approach (P, T, and amounts of the feed) are measured. Then samples are withdrawn from the gas phase. Therefore, small amounts of the substances are directly injected into the carrier gas stream of a gas chromatograph using a pneumatically driven micro sampler (ROLSI: rapid on-line sampler-injector). In the used set-up, where a sapphire tube is placed between two metal flanges, the cell is equipped with two micro samplers (one at the top and one at the bottom of the cell). In order to avoid condensation and adsorption of high boiling components, the micro samplers and the lines for the gas stream are superheated. In order to enable the sampling, the pressure inside the cell has to be larger than the pressure of the carrier gas stream of the gas chromatograph.
In all cases for physical absorption Henry coefficients can be derived from the P-x data.
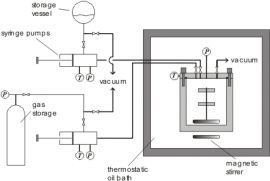
High pressure static synthetic / analytic apparatus
For VLE, vapor pressure, or gas solubility measurements (isothermal P-x data) a static apparatus can be used (see following figures). In our laboratory different designs, e.g., with different cell materials like stainless steel, Hastelloy, titanium, Alloy, or gold plated are available. In this procedure, the pressure of different overall compositions is measured at constant temperature. The apparatus can be operated at temperatures between 200 K and 500 K and pressures up to 20 MPa. The thermostatted, purified and degassed compounds (liquids or liquefied gases) are charged into the VLE cell which is evacuated and placed in a thermostatic oil bath. The pressure inside the cell is monitored with a calibrated pressure sensor. The temperature is measured with a Pt100 resistance thermometer. The total compositions are determined from the known quantities of liquids injected into the equilibrium cell by piston injector pumps. The liquid phase compositions are obtained by solving mass and volume balance equations which are also taking the vapor-liquid equilibrium into account.
For gas solubility measurements with gas mixtures the mentioned analytic-synthetic approach can be applied using a cell with GC sampler for the gas phase.